H2 Organic Chemistry Reaction Summary for Elucidation and Deductive Questions
Follow Ingel on instagram and telegram for FREE study notes and exam hacks!
Looking for JC H2 chemistry tuition?
Call or WhatsApp Ingel at 96726733 now to avoid disappointment!
Comparison of Alcohol, Water, Phenol and Carboxylic Acid Reactivities
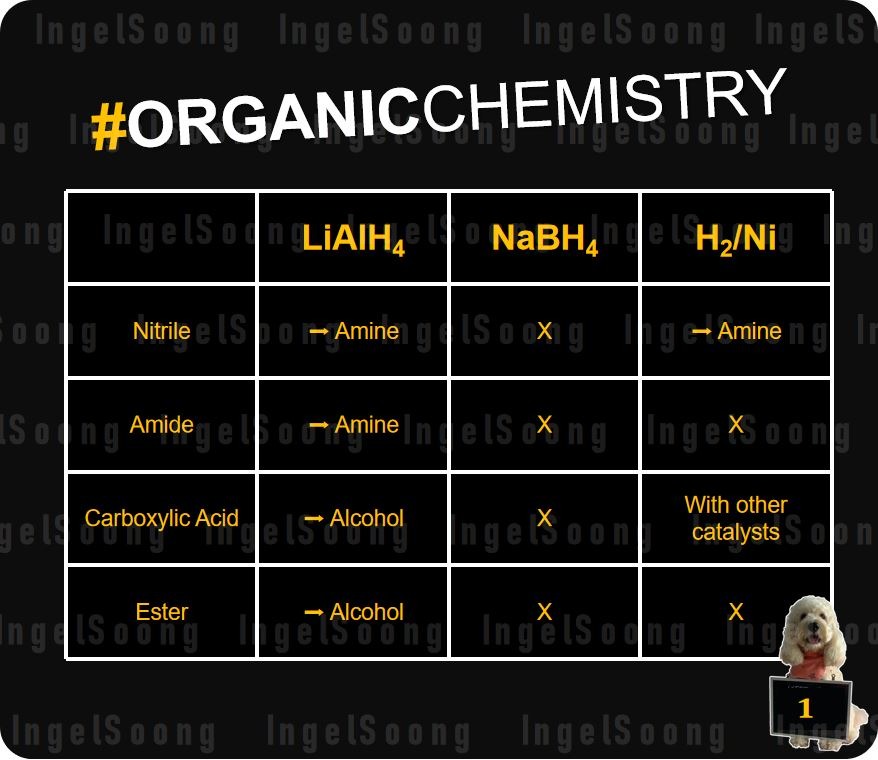
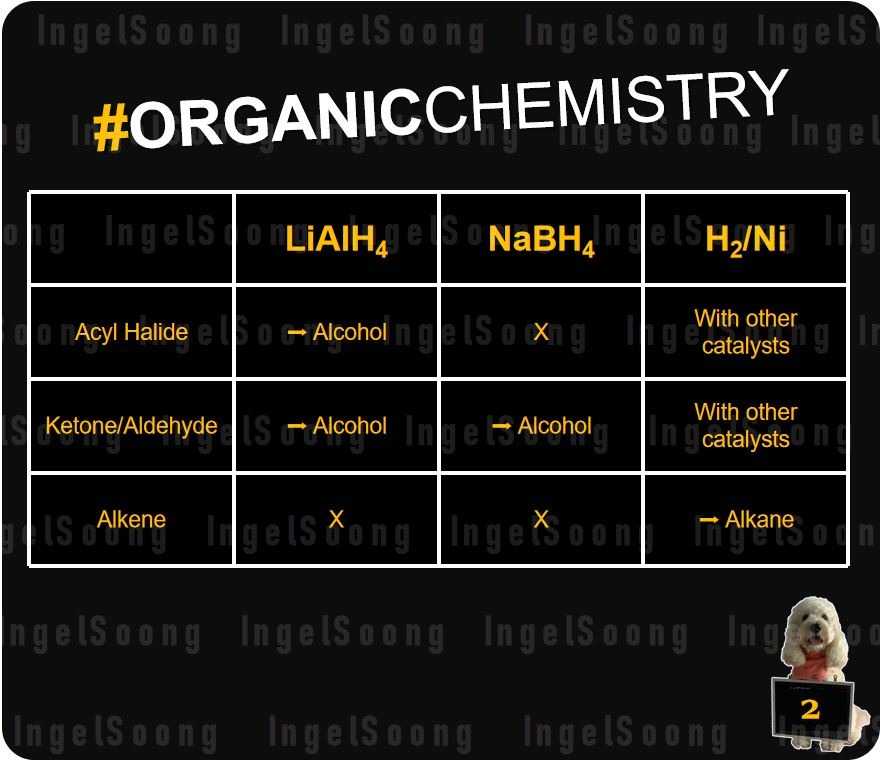
Comparison of LiAl4, NaBH4 and H2 Reduction Reactions
LiAlH4 and NaBH4 works by making the H atom ?-, since the metal atoms are more electronegative. This allows H atom to attack electron deficient C atoms which are bonded to an electronegative atom. They can’t attack alkenes, since alkenes are non-polar.
Comparison of Oxidising Agents
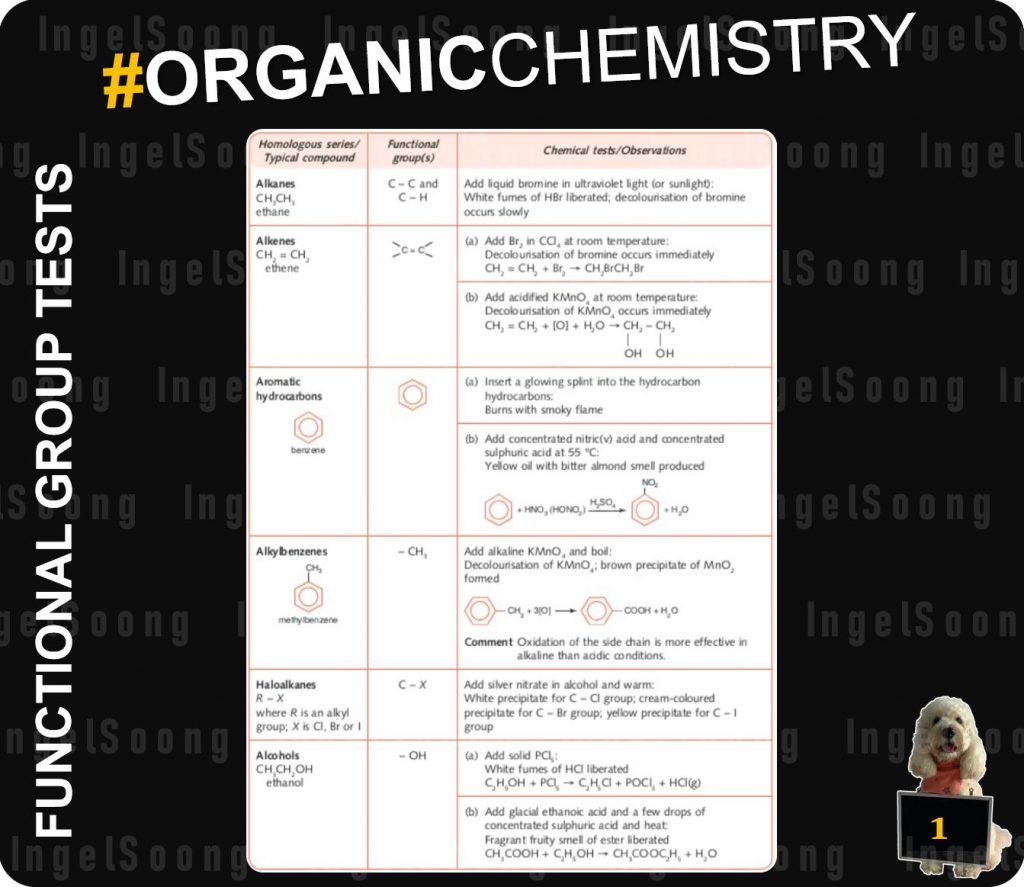
Functional Group Tests
Distinguishing Tests
Reactions with Bromine
Reactions with NaOH
Reactions with H2SO4
Reactions with FeCl3
Reactions with KMnO4
Links
- Reactions of
Alkane, Alkene, Halogenoalkane, Nitrile, Alcohol, Carbonyl Compounds,
Carboxylic Acid, Acyl Chloride, Ester, Nitrogen Compounds
- Formation of
Alkane, Alkene, Halogenoalkane
Nitrile, Alcohol, Carbonyl Compounds
Carboxylic Acid, Acyl Chloride, Ester, Amine, Amide
- Reactions of
Benzene, Methylenzene, Phenol, Aryl Chloride, Aryl Nitro
- Summary map [Aliphatic]
- Summary map [Aromatic]
Aliphatic Compounds
The following is a summary of reaction for hydrocarbons (alkane, alkene, alkyl hydrogensulfate), hydrogen derivatives (halogenoalkane), hydroxyl compounds (alcohol), carbonyl compounds (aldehydes, ketones), carboxylic acids and derivatives (carboxylic acid, acyl chloride, ester), nitrogen compounds (nitrile, amine, amide, amino acid).
Reactions of Alkane
Halogenoalkane
Reagents:
Cl2 gas (or Br2 gas)
Reaction:
Free radical substitution
Observations:
Greenish yellow Cl2 decolorised
(or reddish brown Br2 decolorised)
White fumes of HCl formed
Reactions of Alkene
Halogenoalkane
Reagents:
Cl2 in CCl4 (or Br2 in CCl4)
Reaction:
Electrophilic addition
Observations:
Greenish yellow Cl2 decolorised
(or reddish brown Br2 decolorised)
Diol
Reagents:
Cold dilute KMnO4
Reaction:
Mild Oxidation
Observations:
Purple KMnO4 decolorized.
Brown MnO2 ppt formed in alkaline conditions
Carboxylic Acid
Reagents:
Hot acidified KMnO4
Reaction:
Oxidative cleavage
Observations:
Purple KMnO4 decolorized.
Brown MnO2 ppt formed in alkaline conditions
Carbon dioxide
Reagents:
Hot acidified KMnO4
Reaction:
Oxidative cleavage for terminal alkenes only
Observations:
Purple KMnO4 decolorized.
Reactions of Halogenoalkane
Amine
Reagents:
Excess NH3 (alc), heated in a sealed tube
Reaction:
Nucleophilic substitution
Observations:
–
Reactions of Nitrile
Reactions of Alcohol
Halogenoalkane
Reagents:
PCl3 (or PCl5 or SOCl2)
Conc HCl, anhydrous ZnCl2, heat (for 1o and 2o alcohols only)
PBr3 from Br2 and red P
HBr gas from NaBr and H2SO4, heat
PI3 from I2 and red P
Reaction:
Substitution
Observations:
HCl fumes for PCl3/PCl5/SOCl2 only
Alkoxide ion
Reagents:
Na metal
Reaction:
Reduction
Observations:
Effervescence of H2 gas formed which gives pop sound with lighted splint
Carboxylic acid
Reagents:
1o alcohol only.
KMnO4, heat (or K2CrO4, heat)
Reaction:
Oxidation
Observations:
Purple KMnO4 decolorised
(or orange K2CrO4 turned green)
Aldehyde
Reagents:
1o alcohol only.
K2CrO4, heat with distillation
Reaction:
Oxidation
Observations:
Orange K2CrO4 turned green
Ketone
Reagents:
2o alcohol only.
KMnO4, heat (or K2CrO4, heat)
Reaction:
Oxidation
Observations:
Purple KMnO4 decolorised
(or orange K2CrO4 turned green)
Carboxylate ion
Reagents:
I2, NaOH, heat
Reaction:
Mild oxidation
Observations:
Yellow CHI3 ppt formed
Ester
Reagents:
Acid, conc H2SO4, heat
(or acyl chloride, rtp)
Reaction:
Condensation
Observations:
Sweet smell of ester
Reactions of Carbonyl Compound
Acid
Reagents:
KMnO4, heat (or K2CrO4, heat)
Reaction:
Oxidation
Observations:
Purple KMnO4 decolorised
(or orange K2CrO4 turned green)
Alcohol
Reagents:
NaBH4 (aq), (or LiAlH4 in dry ether, H2 gas, Pt heat)
Reaction:
Reduction
(Aldehyde to 1o alcohol
Ketone to 2o alcohol)
Observations:
–
2,4-DNPH
Reagents:
2,4-DNPH, warm
Reaction:
Condensation
Observations:
Orange ppt of hydrazone formed
Carboxylate ion
Reagents:
Fehling’s solution (Cu2+ and NaOH), warm
Aldehyde (except benzaldehyde) only
Reaction:
Oxidation
Observations:
Brick red of Cu2O ppt formed
Carboxylate ion
Reagents:
Tollen’s reageant (Ag2O in NH3)
Aldehyde only
Reaction:
Oxidation
Observations:
Silver mirror of Ag formed
Reactions of Carboxylic Acid
Carboxylate ion
Reagents:
Na metal
Reaction:
Reduction
Observations:
Effervescence of H2 gas formed which gives pop sound with lighted splint
Carboxylate ion
Reagents:
Na2CO3 (or NaHCO3)
Reaction:
Neutralisation
Observations:
Effervescence of CO2 gas formed which gives white ppt with limewater
Acyl chloride
Reagents:
PCl3 (or PCl5 or SOCl2)
Reaction:
Neutralisation
Observations:
White fumes of HCl
Carbon dioxide
Reagents:
KMnO4, heat
HCOOH or (COOH)2
Reaction:
Purple KMnO4 decolorized.
Effervescence of CO2 gas formed which gives white ppt with limewater
Observations:
–
Reactions of Acyl Chloride
Amide
Reagents:
Excess conc NH3
Excess RNH2
1o amine to 2o amide
2o amine to 3o amide
no reaction for 3o amide
Reaction:
Condensation
Observations:
White fumes of HCl
Reactions of Ester
Reactions of Nitrogen Compound
Alkyl ammonium and carboxylic acid
Reagents:
HCl(aq)
(or CH3COOH)
Reaction:
Neutralisation
Observations:
White crystalline solid
Alkyl ammonium
Reagents:
Dilute H2SO4, heat under reflux
Reaction:
Acid hydrolysis
1o amide produce NH4+
Observations:
–
Carboxylate ion and amine
Reagents:
Dilute NaOH, heat under reflux
Reaction:
Base hydrolysis
1o amide produce NH3
Observations:
NH3 gas turned damp red litmus paper blue
Formation of Alkane
Formation of Alkene
Formation of Halogenoalkane
Alkene
Reagents:
Cl2 in CCl4 (or Br2 in CCl4)
Reaction:
Electrophilic addition
Observations:
Greenish yellow Cl2 decolorised
(or reddish brown Br2 decolorised)
Alkene
Reagents:
PCl3 (or PCl5 or SOCl2)
Conc HCl, anhydrous ZnCl2, heat (for 1o and 2o alcohols only)
PBr3 from Br2 and red P
HBr gas from NaBr and H2SO4, heat
PI3 from I2 and red P
Reaction:
Substitution
Observations:
HCl fumes for PCl3/PCl5/SOCl2 only
Formation of Alcohol
Halogenoalkane
Reagents:
NaOH (aq), heat under reflux
Reaction:
Nucleophilic substitution
Observations:
–
Carbonyl Compound
Reagents:
NaBH4 (aq), (or LiAlH4 in dry ether, H2 gas, Pt heat)
Reaction:
Reduction
(Aldehyde to 1o alcohol
Ketone to 2o alcohol)
Observations:
–
Formation of Carboxylic Acid
Alkene
Reagents:
Hot acidified KMnO4
Reaction:
Oxidative cleavage
Observations:
Purple KMnO4 decolorized.
Brown MnO2 ppt formed in alkaline conditions
Alcohol
Reagents:
1o alcohol only.
KMnO4, heat (or K2CrO4, heat)
Reaction:
Oxidation
Observations:
Purple KMnO4 decolorised
(or orange K2CrO4 turned green)
Formation of Acyl Chloride
Carboxylic acid
Reagents:
PCl3 (or PCl5 or SOCl2)
Reaction:
Neutralisation
Observations:
White fumes of HCl
Formation of Ester
Alcohol
Reagents:
Acid, conc H2SO4, heat
(or acyl chloride, rtp)
Reaction:
Condensation
Observations:
Sweet smell of ester
Carboxylic acid
Reagents:
Alcohol, conc H2SO4, heat
Reaction:
Condensation
Observations:
Sweet smell of ester
Acyl Chloride
Reagents:
ALcohol, conc H2SO4, heat
Reaction:
Condensation
Observations:
Sweet smell of ester
Formation of Amine
Halogenoalkane
Reagents:
Excess NH3 (alc), heated in a sealed tube
Reaction:
Nucleophilic substitution
Observations:
–
Amide
Reagents:
Dilute NaOH, heat under reflux
Reaction:
Base hydrolysis
1o amide produce NH3
Observations:
NH3 gas turned damp red litmus paper blue
Formation of Amide
Acyl chloride
Reagents:
Excess conc NH3
Excess RNH2
1o amine to 2o amide
2o amine to 3o amide
no reaction for 3o amide
Reaction:
Condensation
Observations:
White fumes of HCl
Aromatic Compounds
The following is a summary of reaction for hydrocarbons (benzene), hydroxyl compounds (phenol), carboxylic acids and derivatives (benzoic acid, benzoate), nitrogen compounds (phenylamine, nitrobenzene):
Reactions of Benzene
Nitrobenzene
Reagents:
Conc HNO3, conc H2SO4, heat under reflux at 55oC
Reaction:
Electrophilic substitution
Observations:
Yellow layer of nitrobenzene formed
Bromobenzene
Reagents:
Br2, anhydrous FeBr3, rtp
(or Cl2, anhydrous FeCl3, rtp)
Reaction:
Electrophilic substitution
Observations:
Reddish brown Br2 decolorised
(or greenish yellow Cl2 decolorised)
Reactions of Methylbenzene
1-methyl,2-nitrobenzene
Reagents:
Conc HNO3, conc H2SO4, heat under reflux at 30oC
Reaction:
Electrophilic substitution
Observations:
–
1-bromo-2-nitrobenzene
Reagents:
Br2, anhydrous FeBr3, rtp
(or Cl2, anhydrous FeCl3, rtp)
Reaction:
Electrophilic substitution
Observations:
Reddish brown Br2 decolorised
(or greenish yellow Cl2 decolorised)
chloromethylbenzene
Reagents:
Cl2, UV light, boil
Reaction:
Free radical substitution
Observations:
Greenish yellow Cl2 decolorised
White fumes of HCl formed
Benzoic acid
Reagents:
Hot acidified KMnO4, heat under reflux
Reaction:
Side chain oxidation
Observations:
Purple KMnO4 decolorised
White benzoic acid ppt formed
Reactions of Phenol
Phenoxide ion
Reagents:
Na metal
Reaction:
Reduction
Observations:
Effervescence of H2 gas formed which gives pop sound with lighted splint
Phenyl benzoate
Reagents:
CH3Cl, pyridine, rtp
Reaction:
Condensation
Observations:
Sweet smell of ester
White fumes of HCl formed
Phenyl benzoate
Reagents:
Phenylacetyl chloride, NaOH(aq), rtp
Reaction:
Condensation
Observations:
Sweet smell of ester
White fumes of HCl formed
1,3,5-tribromobenzene
Reagents:
Br2(aq), rtp
Reaction:
Condensation
Observations:
Reddish brown Br2 decolorized
White 1,3,5-tribromobenzene ppt formed
White fumes of HBr formed
Nitrophenol
Reagents:
Dilute HNO3(aq), rtp
Reaction:
Electrophilic substitution
Observations:
Yellow layer of nitrophenol formed
1,3,5-trinitrophenol
Reagents:
Conc HNO3(aq), rtp
Reaction:
Electrophilic substitution
Observations:
Yellow ppt of 1,3,5-trinitrophenol formed
1,3,5-trinitrophenol
Reagents:
Neutral FeCl3(aq)
Reaction:
Electrophilic substitution
Observations:
Violet complex compound formed
Reactions of Aryl Chloride
Phenyl benzoate
Reagents:
Phenol, NaOH(aq), rtp
Reaction:
Condensation
Observations:
Sweet smell of ester
White fumes of HCl formed
Reactions of Aryl Nitro Compounds
Phenylamine
Reagents:
Sn, conc HCl, heat followed by NaOH (or H2, Ni, heat)
Reaction:
Reduction
Observations:
Sweet smell of ester
White fumes of HCl formed
2,4,6-tribromophenylamine
Reagents:
Br2(aq)
Reaction:
Electrophilic substitution
Observations:
Reddish brown Br2 decolorized
White 2,4,6-tribromophenylamine ppt formed
White fumes of HBr formed
































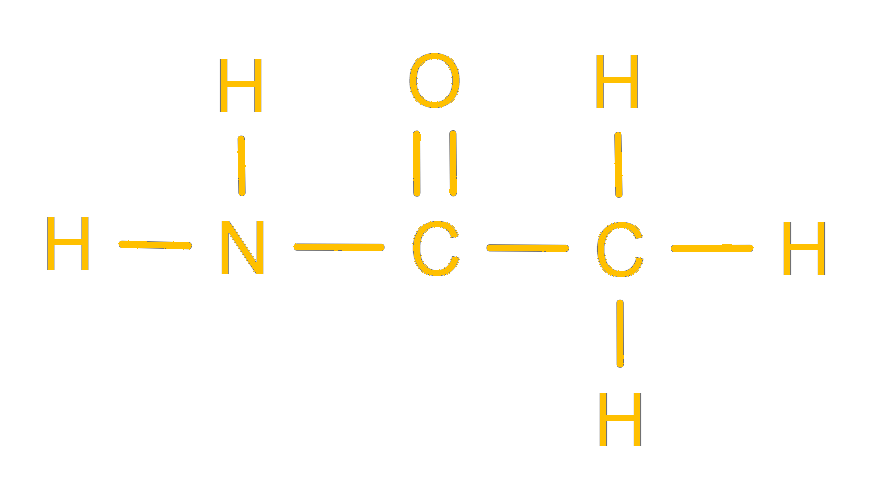

























This is certainly pleasing to the eyes. Thank you for making the dreaded organic chemistry much more appealing
This comes in handy when I needed it most.