Electrochemistry
Here’s an “ingelious” way to solve redox questions related to electrochemistry.
Let’s look at a typical JC A Level H2 Chemistry question:
Determine if the following reaction is spontaneous:
$Cr_2O_7^{2−}+2I^−➡ Cr^{3+}+I_2$
Say we want to form a complete picture of the reaction.
Step 1: Write down the $E^°$ values of both reactants.
Step 2: From the oxidation states, determine which reactant undergo oxidation, which reactant undergo reduction.
Step 3: Determine if the reaction can happen by calculating the $E_{cell}^°$. Assume the reaction takes place first and never change the sign of the half-equations.
Step 4: The electrode where oxidation takes place is always the anode. The electrode where reduction takes place is always the cathode. This is true for both electrolytic cell and electrolysis.
Step 5: Determine the direction of electron flow by checking which electrode undergoes oxidation. See how electrons are produced from the equation below:
$M ➡ M^{+}+e^−$ (anode)
Step 6: The electrode that produces electrons is the negative electrode. Never memorise anode being the negative electrode, since it will change depending on the cell being an electrolytic one or electrolysis is taking
place.
Say we want to form a complete picture of the reaction.
Step 1: Write down the E^° values of both reactants.
$Cr_2O_7^{2−}+14H^++6e^−➡ Cr^{3+}+7H_2O $ +1.33V
$I_2+2e^−➡ 2I^− $ +0.54V
Step 2: From the oxidation states, determine which reactant undergo oxidation, which reactant undergo reduction.
Cr in $Cr_2O_7^{2−}$ is +6, reduced to +3 in $Cr^{3+} $
$I^−$ is -1, oxidised to 0 in$ I_2 $
Step 3: Determine if the reaction can happen by calculating the $E_{cell}^°$. Assume the reaction takes place first and never change the sign of the half-equations.
$ E_{cell}^° = E_{red}^° −E_{oxi}^° $
= +1.33 – (+0.54)
= +0.79V
Step 4: The electrode where oxidation takes place is always the anode. The electrode where reduction takes place is always the cathode. This is true for both electrolytic cell and electrolysis.
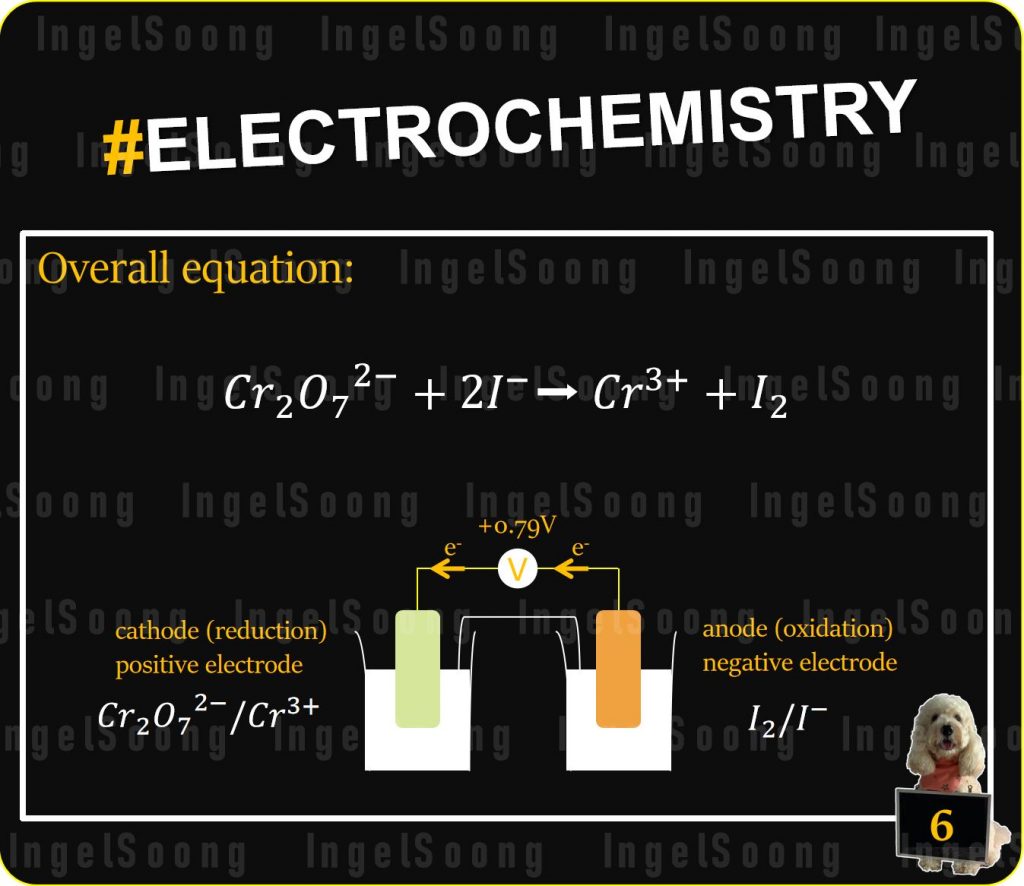
$Cr_2O_7^{2−} $ electrode is the cathode
$I^− $ electrode is the anode
Step 5: Determine the direction of electron flow by checking which electrode undergoes oxidation. See how electrons are produced from the equation below:
$I_2+2e^−➡ 2I^−$
Electrons flow from $I_2 $ electrode to $Cr_2O_7^{2−}$ electrode
Step 6: The electrode that produces electrons is the negative electrode. Never memorise anode being the negative electrode, since it will change depending on the cell being an electrolytic one or electrolysis is taking place.
$I_2$ electrode is the negative electrode
Overall equation: $Cr_2O_7^{2−}+2I^−➡ Cr^{3+}+I_2$
Let’s look at another typical JC A Level H2 Chemistry question:
Determine if the following reaction is spontaneous:
$Cr_2O_7^{2−}+2Cl^−➡ Cr^{3+}+Cl_2$
Say we want to form a complete picture of the reaction.
Step 1: Write down the E^° values of both reactants.
$Cr_2O_7^2−+14H^++6e^−➡ Cr^{3+}+7H_2O$ +1.33V
$Cl_2+2e^−➡ 2Cl^−$ +1.36V
Step 2: From the oxidation states, determine which reactant undergo oxidation, which reactant undergo reduction.
Cr in $Cr_2O_7^2−$ is +6, reduced to +3 in $Cr^{3+} $
$Cl^−$ is -1, oxidised to 0 in $Cl_2$ (remember, you do not have $Cl_2$ to begin with)
Step 3: Determine if the reaction can happen by calculating the $E_{cell}^°$. Assume the reaction takes place first and never change the sign of the half-equations.
$E_{cell}^° = E_{red}^° −E_{oxi}^°$
= +1.33 – ( +1.36)
= -0.03V (non-spontaneous)
For electrolysis, the non-spontaneous reaction is driven by an external battery, which is essentially the electrolytic cell.
Purpose of salt bridge:
1. To complete the circuit so that electrons can flow.
2. To maintain electrical neutrality in each cell. Otherwise the reactions will stop shortly after they start. (If there are many $Zn^2+ $in the right half-cell, electrons are harder to be removed due to electrostatic attraction, and current will cease to flow).